"Hi doctor, could you prescribe me that once-weekly injection for weight loss? It can work for my diabetes too, right?" Mr T asks during one of his regular reviews.
If you have not already faced this scenario, there is a good chance that you will someday soon. Thanks to the weight loss effect of once-weekly incretin-based therapies such as semaglutide and tirzepatide, a disease-centric approach to the management of type 2 diabetes mellitus (T2D) has now become more attainable, and patients are initiating requests for these therapies. This well-established approach recognises that the physiological mechanisms behind obesity and T2D are inextricably intertwined, and thus recommends treating obesity not merely as a risk factor but as the primary therapeutic target in T2D. This is all well and good for people with obesity (PwO) who also have diabetes, but what if Mr T had obesity without diabetes? Should that undermine the treatment of his obesity?
Incretin-based therapies
The year 2021 welcomed a game changer in the form of semaglutide for treatment of T2D in Singapore: Once-weekly subcutaneous (SC) semaglutide 1 mg (Ozempic®) and daily oral (PO) semaglutide (Rybelsus®). It was not until February 2023 that once-weekly SC semaglutide 2.4 mg (Wegovy®) was approved for obesity treatment, and in the following month, tirzepatide (Mounjaro®) received approval for the treatment of T2D. Most recently on 8 November 2023, the US Food and Drug Administration approved through an expedited process the use of tirzepatide (Zepbound®) for chronic weight management in adults with obesity, reflecting the urgent need to address gaps in obesity treatment.
These newer incretin-based therapies are revolutionary in combining superior glucose-lowering effects with unprecedented weight loss (see Tables 1 and 2). This contrasts with conventional T2D therapies like sulphonylureas, insulin and thiazolidinediones, which induce weight gain as a by-product of treatment. Essentially, glucagon-like peptide-1 (GLP-1) receptor agonists (RA) and glucose-dependent insulinotropic polypeptide (GLP-1/GIP) dual RA target the fundamental pathophysiology that underpins both T2D and obesity. This is achieved by virtue of their metabolic effects and reduction of body weight through their central actions of increased satiety and reduced food intake (see Figure 1), thus rendering them highly effective therapies. In addition, they only require once-a-week dosing and have mild side-effect profiles. It is no wonder that more patients like Mr T are now taking the initiative to request these treatments.
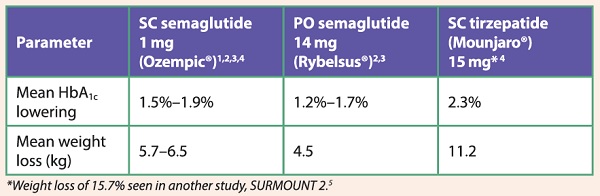
Table 1: Mean HbA1c and weight changes in patients with T2D treated with semaglutide and tirzepatide
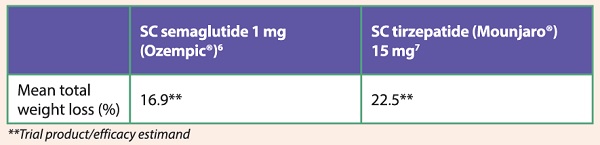
Table 2: Weight loss in patients with obesity (without T2D) treated with semaglutide and tirzepatide
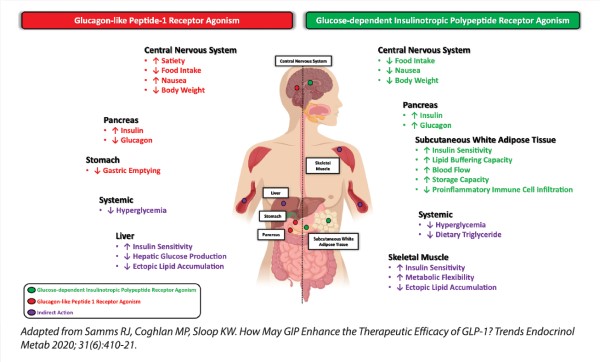 Figure 1: Pleiotropic effects of dual GIP/GLP-1 RA
Figure 1: Pleiotropic effects of dual GIP/GLP-1 RA
Influence of social media (over doctors'!)
Semaglutide's skyrocketing popularity has been fuelled by social media. On TikTok for example, the hashtag #Ozempic has garnered over 1.3 billion views while #Mounjaro boasts an impressive 819 million views since its recent launch in 2022. Social media influencers have been enthusiastic in sharing their personal experiences with semaglutide, and the pervasiveness of their influence has had real implications on drug availability across the globe. Such unregulated and nondiscriminatory word-of-mouth marketing gives it legitimacy in the eyes of the public and has broken down barriers to the use of these medications, as such forms of messaging are seen as more relatable than clinical trials. While we physicians need to repeatedly present our patients with scientific data to convince them to adopt even the most effective of medical treatments, social media upstages us with a fraction of the effort.
Controversies and consequences
In most parts of the world including Singapore, Wegovy® is not available for clinical use due in part to a global supply shortage. The surge in demand for semaglutide has channelled the off-label use of Ozempic® and Rybelsus® for weight management, further straining the supply of these medications for use in T2D. In the US alone, prescriptions for incretin-based treatments for obesity (Ozempic®, Mounjaro®, Rybelsus® and Wegovy®) rose approximately 260% from 2021 to 2022, and 25% of these were in patients without diabetes.8
Some have argued that such off-label use deprives patients with T2D of essential treatments. The counter-argument may be that patients with T2D have more therapeutic options, some of which have proven additional mortality and cardiorenal benefits.9,10 Additionally, some proponents for the use of Wegovy® for obesity treatment posit that it is essentially the same medication as Ozempic®, just in a different pen device which allows for delivery of higher doses. For example, the up-titration of semaglutide 2.4 mg (Wegovy®) requires patients to receive 1 mg of semaglutide (ie, Ozempic® doses) for at least four weeks and this may be an adequate dose for some patients.
Furthermore, our obesogenic environment poses an insurmountable obstacle to weight loss without professional support for most PwO. More than four in five PwO have made a serious attempt at weight loss but less than half of them would have lost 5% of their body weight over three years, and 70% of those that do are expected to regain their losses.11 This illustrates that regardless of the drivers of obesity, PwO are in an established state of pathological energy homeostasis that requires intensification of treatment beyond foundational lifestyle changes, and which often requires the use of anti-obesity medications (AOMs).
In particular, PwO with severe obesity-related complications (ORC), such as heart failure or fibrosis from steatotic liver disease, should not have their treatment delayed. Expedited and substantial weight loss, which often necessitates AOM, is required to halt disease progression, improve the severity of symptoms or even send some of these ORC into remission. Unfortunately, the history of AOM has been fraught with the abrupt withdrawal of several medications due to adverse effects (eg, sibutramine, lorcaserin, rimonabant and fenfluramine-phentermine). Though we have effective AOM in our armamentarium, such as phentermine, extended-release naltrexone and bupropion, and liraglutide, their more modest weight loss (mean total 4% to 8%)1 and/or side-effect profiles often preclude their use. On the other hand, semaglutide has been shown in clinical trials to be well tolerated, safe and highly beneficial to patients with established cardiovascular disease,12 heart failure13 and other ORC, but without T2D.1 SC semaglutide is thus a potentially life-saving drug for PwO even in the absence of pre-existing T2D.
What is absolutely not acceptable is the off-label use by people who do not meet the clinical criteria for AOM. Some have resorted to unregulated sources to get a quick fix without consulting a healthcare professional trained in obesity management, with no screening of eligibility and contraindications, and without monitoring for efficacy or side effects. Doctors have at times been accomplices by recklessly offering prescriptions for these medications with minimal or no patient assessment and support. Such practices have opened the gateway for considerable potential for abuse.
Semaglutide's popularity in the media has proven to be a double-edged sword. It was inevitable for it to start making headlines for the wrong reasons: from reports of gastroparesis, suicide attempts and critical supply shortages to unflattering excess skin after weight loss. While the actual numbers reported with these "serious" medical side effects are small and not currently substantiated, these reports have caused a media frenzy. The fear of these exaggerated side effects could lead to treatment aversion in PwO who truly need it. Judicious use of semaglutide through appropriate patient selection and vigilance can mitigate some of its more serious side effects such as serious gastrointestinal symptoms, gallstone pancreatitis and exacerbation of depression. Proper public and patient education on the holistic management of obesity, complemented with the appropriate use of AOM, is needed to counteract the unscientific media sources.
Perhaps the biggest controversy stems from the perception we – doctors, healthcare professionals, policymakers and the general public – have of obesity. When faced with a shortage of semaglutide, some countries have opted to prioritise treatment of T2D, resulting in the delay or discontinuation of the treatment for PwO. This is despite the evidence that semaglutide is effective in both conditions; its effect on glucose metabolism is equally strong as its appetite-modulating effect. Discontinuing GLP-1 RA leaves our patients unsupported in their battle against the inevitable weight regain that occurs as a consequence of physiological forces, such as an increase in appetite and reduction in metabolic rate. Such policy decisions ignore the pathophysiology of obesity. Thus, the decision to view semaglutide primarily as a "diabetic medication" goes beyond just semantics – it reveals our subtle biases and has tangible downstream implications.
Treatment of obesity leads to the prevention of metabolic, mechanical and psychological obesity complications, and not to mention, also leads to an improvement in patients' quality of life and a reduction in healthcare spending and mortality. So why are we putting the cart before the horse by not prioritising the treatment of obesity? Why is it then that when push comes to shove, PwO must "sacrifice" a treatment which has greatly benefitted their health or forgo one that could be life-saving? Beyond the weight regain experienced with cessation of effective anti-obesity treatment, PwO will experience a worsening of health with the relapse or deterioration of obesity-related complications, the deepening of weight stigma and a reduced quality of life.
Final thoughts
The superior efficacy of GLP-1 RA and newer incretin-based therapies on glycaemic control in T2D and weight loss in obesity is undeniable, and there are many more promising therapies on the horizon. Recent phase two studies of retatrutide, a GLP-1/GIP/glucagon triple RA, showed a similar glucose-lowering effect,14 but an even greater weight loss effect (24.2% at 48 weeks) than tirzepatide.15
It is not our intention to advocate the use of AOM, in particular GLP-1 RA, for every patient with obesity. The cost of the newer GLP-1 RA runs high in countries where they are available. Since treatment of obesity as a chronic disease would require long-term AOM use in many individuals, the cost-effectiveness of this approach has yet to be established and such an approach, while clinically recommended, may potentially put a strain on health systems. At present, obesity prevalence is predicted to rise to impact about one in four of the world's population in 2035. We urgently need to implement a framework of risk stratification which goes beyond the use of body mass index cutoffs, prioritising people with more severe ORC in the treatment paradigm who would benefit the most from efficacious AOM like incretin-based therapies and in whom we expect treatment to be more cost-effective. This priority system should be on par with the priority we allot to people with T2D.
Formalised and updated education on the scientific paradigm of diabetes and obesity will help more to understand the pathophysiology of these two diseases. A better understanding is urgently needed to address the root cause of obesity stigma, which manifests in our misperceptions of obesity.16 This understanding must lead to definitive actions that provide equality to the treatment of obesity within a chronic disease framework, one in which PwO will not be discriminated against and will be equally supported.
With more novel therapies targeting both obesity and T2D in the pipeline, this is only the beginning of our discussion. Once we acknowledge obesity as a disease in and of itself – a pathophysiological condition – we will move beyond having to make justifications for the use of efficacious AOM for PwO. The earlier we intervene on the precursor disease of multiple diseases, the less PwO need to suffer from the burdensome sequalae of obesity and the better the quality of life our patients with obesity can have.
Disclosure: Dr Tham reports receiving honoraria for speaking engagements for Novo Nordisk, iNova, EuroDrug Laboratories and DKSH, and engagement in scientific committees and advisory boards for Novo Nordisk and DKSH.
